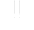Arthur N. Palmer and Margaret V. Palmer
Department of Earth Sciences, State University of New York ,
Oneonta.NY 13 820-4015, USA
Abstract
Low-velocity capillary seepage in the vadose zone is responsible for a variety of geochemical processes in Mammoth Cave . Water that infiltrates through the cap-rock of detrital sandstone and shale is isolated from the high-CO 2 of the soil before it encounters the underlying carbonate rocks, so that carbonate dissolution in narrow fissures around the cave takes place under nearly closed conditions with respect to CO 2 . As a result, the equilibrium P CO2 of the capillary water decreases to nearly zero and the pH can rise to more than 9.0. When the water emerges into the cave it rapidly absorbs CO 2 from the cave air and becomes highly aggressive toward carbonate rocks. Where discrete trickles exit from fissures, deep irregular rills are formed. Where the flow is more diffuse, the cave walls are weathered to a chalky white by partial dissolution and recrystallization of the carbonate rock. If the water has acquired sulfate from oxidation of pyrite or dissolution of residual gypsum within the bedrock, the SO 4 = /CO 3 = ratio of the water rises sharply at the cave walls, promoting the replacement of carbonate bedrock by gypsum. The pH decrease caused by the uptake of CO 2 enables silica to precipitate in small amounts in the weathering rind. Direct measurement of capillary water chemistry is difficult because of the small quantity and inaccessibility of the water involved, but it can be reliably inferred from the geochemical setting and the effects upon the cave.
Keywords: capillary seepage, geochemical processes in vadose zone, limestone weathering in caves
Introduction
Vadose water in the Mammoth Cave System spans a broad range of flow rates, chemistry, and relation to local geologic conditions. At the high-discharge end of the spectrum, vadose streams fed by concentrated inputs such as sinking streams or infiltration retain their solutional aggressiveness over large distances and are responsible for the origin of cave passages. Smaller vadose trickles and seeps tend to be close to saturation with dissolved carbonates and account for the rather sparse travertine deposits in the cave where CO 2 degassing takes place. At the low-discharge extreme is capillary seepage, in which the movement is governed mainly by cohesive and adhesive forces. The walls of dry passages possess a very low capillary potential in relation to that of the moist bedrock around the cave, and this potential gradient draws water toward the caves from all directions, even against gravity.
Capillary water accounts for a variety of unexpected geochemical and mineralogical effects, which, although volumetrically slight, have a profound effect on the appearance of the cave walls. They also provide insight into the patterns of vadose recharge to the cave and the geochemistry of bedrock weathering.
Field observations
It is well known that capillary water transports dissolved solids to the cave walls, and that deposits such as gypsum, aragonite, and some varieties of cave popcorn result from evaporation and CO 2 degassing at the cave walls. This water is rarely visible except for a thin static film of moisture. However, discrete flows of capillary seepage can be seen in many places in Mammoth Cave where water emerges from tiny fissures that have no visible solutional enlargement. At the exit point the water is at (or very near) saturation with respect to the carbonate minerals in the bedrock, as shown by the lack of solutional enlargement of the openings through which it issues, and by direct measurement of drips in other similar caves. This is to be expected of capillary seepage because of the long residence time and intimate contact with the bedrock. However, below the inflow points, where the water seeps down the cave wall, it has dissolved prominent rills with depths as much as 1-2 cm. Evidently the water suddenly acquires a great deal of solutional aggressiveness by the rapid uptake of CO 2 from the cave atmosphere.
This is strange behavior, because the Pco 2 of the cave atmosphere is much lower than that of the overlying soil through which the water infiltrates, Measurements of soil CO 2 by Miotke (1975) in various places in the Chester Upland over Mammoth Cave, over a variety of seasons, show a mean Pco 2 of 0.005 atm, with typical values of 0.001 atm in the sandy, well-aerated soils of the sandstone-capped ridges. These values are low in comparison with most other soils in humid climates. For example, Miotke's CO 2 measurements in the less permeable, clay-rich soils of the Pennyroyal Plateau average 0.012 atm. His measurements of air in Mammoth Cave show an extremely low mean value of 0.0006 atm, which is not even twice that of the surface atmosphere (about 0.00035 atm). Air in Mammoth Onyx Cave , in the Pennyroyal Plateau near Horse Cave , has a mean value twice as great (Miotke, 1975).
The equilibrium Pco 2 in standing pools in Mammoth Cave is perhaps an even more reliable indicator of long-term CO 2 values toward which capillary seepage trends when it enters the cave. Our measurements of the chemistry of standing vadose pools in Mammoth Cave show a mean value of 0.0009 atm. Microbial activity may account for the slight elevation of CO 2 in the pools in comparison with that of the cave air.
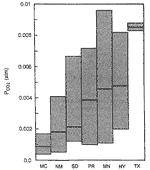
The Pco 2 of the atmosphere in Mammoth Cave and others in the Chester Upland is unusually low in comparison with nearly all other caves, even those in semi-arid regions (Fig.1). These values were determined from equilibrium calculations using our chemical measurements of cave pools in various parts of the country. Mammoth Cave is by far the lowest in both mean Pco 2 (0.00088 atm) and standard deviation (0.00048 atm). Locations of pools sampled in Mammoth Cave include the Cleaveland-Boone Avenue area, many locations in Crystal Cave , and several in Pohl Avenue , Turner Avenue , and the Overlook area. The second-lowest group shown in Fig.1, from the Guadalupe Mountains of New Mexico , has a mean Pco 2 nearly twice as great.
Fig. 1. Pco 2 of standing pools in Mammoth Cave , compared to that of other caves. Shaded boxes show range of measured values, with mean values shown by heavy lines. MC = Mammoth Cave (N = 11); NM = Carlsbad Cavern and Lechuguilla Cave, New Mexico (N = 17); SD = Jewel, Wind, and Reed's Caves, South Dakota (N = 6); PR = Sistema Rio Encantado, Puerto Rico (N = 17); MN = Mystery Cave, Minnesota (N = 49); NY = McFail's and Barrack Zourie Caves, New York (N = 10); TX = Sonora Caverns, Texas (N = 2).
The pH and Pco 2 of seepage and drips entering the cave can indicate conditions higher in the vadose zone. To do so, pH must be measured at the exact point where the water enters, before it has had an opportunity to begin equilibration with CO 2 in the cave atmosphere. This is rarely possible, because the inflow points are usually inaccessible or consist of passages too small to traverse, in which equilibration with the cave air has already begun. Vadose drips typically lose CO 2 so rapidly that their pH increases at least 0,5 pH unit before they hit the floor. Even a slight discrepancy in pH can greatly affect the equilibrium calculations. The change in pH of emerging capillary water is probably just as fast. In the few places where we have been able to measure vadose drips at the input sites, they have been exactly at saturation with dissolved calcite, which shows that the water has fully equilibrated with the limestone by the time it reaches the cave. We have not yet been able to measure the chemistry of capillary seepage in Mammoth Cave , owing to the small quantity of flow. However, the overall chemical pattern is clear from the known Pco 2 of the soil and cave air, and from the geochemical effects of the water on the cave.
Since the soil and vadose drips have a much higher Pco 2 than that of the cave air, how is it possible for the CO 2 level of the capillary seepage within the limestone around Mammoth Cave to be lower than that of the cave? The answer must lie in the nature of the dissolution process in the narrow fissures that transmit the water.
Closed-system dissolution in the vadose zone
In a typical karst region of soil-mantled limestone, water that infiltrates through the soil approaches saturation with dissolved carbonates while still in contact with abundant CO 2 , even at considerable depth below the surface (Atkinson, 1977). Although the dissolution of limestone consumes CO 2 in the water, it is rapidly replenished by CO 2 from the overlying soil. However, in the Chester Upland, where much of the infiltrating water passes through thick quartz sandstone before encountering limestone, the CO 2 consumed by carbonate dissolution cannot be easily replenished from the soil. In the tiny fissures and pores containing capillary water, this process takes place in conditions that are essentially closed with respect to CO 2 . Therefore the CO 2 level in the water drops nearly to zero, the pH rises to unexpectedly high values (often well above 9.0), and the water reaches saturation with limestone at extremely low values (less than 0.2 millimole/liter, equivalent to only 10-15 ppm CaCO 3 ). This process is valid whether or not the first carbonates are encountered beneath the sandstone or interbedded within it.
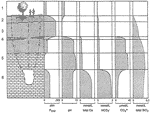
When such water emerges into the cave, CO 2 is rapidly absorbed from the cave air. The equilibrium Pco 2 of the water rises and the pH and carbonate saturation ratios drop sharply. The limestone walls are vigorously dissolved, and the water approaches saturation with carbonate minerals at the new and much higher Pco 2 . Fig.2 shows the probable geochemical evolution of vadose seepage under the typical Pco 2 conditions of soil and cave air in the Mammoth Cave System. Ionic concentrations in Fig.2 were derived from computer-aided equilibrium calculations (see Palmer, 1991). The radical shifts in pH and Pco 2 shown in Fig.2 are most pronounced where the soil CO 2 is rather low, as is the case in the Chester Upland. The importance of the geologic setting is clear.
Fig. 2. Chemical evolution of capillary seepage in the Mammoth Cave area. 1: Rainfall absorbs small amounts of CO 2 from the atmosphere. 2: Infiltrating water acquires greater Pco 2 from soil; pH drops to 4.5-5.0. 3: Seepage water in sandstone becomes isolated from the CO 2 in the soil. 4, 5: Water dissolves limestone in a nearly closed system; Pco 2 drops almost to zero; pH rises to more than 9.0; CO 3 = and silica solubility rise. 6: Seepage water enters the cave and acquires CO 2 from the cave atmosphere; pH, CO 3 , and silica solubility drop sharply; gypsum replaces calcite in evaporative zones. Note: CO 3 = concentration is in micromoles/liter. Silica equilibrium is rarely achieved, so SiO 2 concentrations are crude estimates.
The rills below the outflow points of capillary seepage are irregular and braided, with rough, hackly surfaces. These characteristics show that the flow of solvent water is governed not only by gravity but by surface tension, with low velocity and film thickness, but that the water is also highly undersaturated with calcite. Despite the low CO 2 content of the cave air, the concentration of dissolved calcite increases as much as 3 to 6 times over the initial concentration where the seepage enters. The best examples are in passages that lie beneath a thin sandstone cap along ridge margins (e.g., Boone Avenue in Mammoth Cave ).
This phenomenon may also occur in a few places at the surface. Field notes describing limestone outcrops and quarries in Indiana by Richard L. Powell ( Bloomington , Indiana , personal communication) include references to what he calls "zig-zags," which are solutional grooves wandering erratically for short distances across the rock faces.
The unusually low Pco 2 of the air in Mammoth Cave is caused by several factors, including the rather low values in the granular well-aerated soil, the limited amount of vadose recharge, and ample air exchange with the surface. However, other caves have greater air exchange and similarly low vadose infiltration through the soil, and yet have rather high CO 2 levels. It is possible that the uptake of CO 2 , by capillary seepage lowers the Pco 2 of the cave air, although quantifying this phenomenon would be problematic.
It is very difficult to measure the chemical behavior of capillary seepage, because the closed-system dissolution takes place in inaccessibly tiny fissures and pores. The mere act of exposing the water to measurement by conventional methods (in drill holes, for example) would destroy the very closed system that we are attempting to document. That is why this kind of geochemical system has been overlooked in the past. It has been invoked only a few times before to explain certain conditions of cave origin (Palmer and Palmer, 1989; Palmer, 1991). We have had marginal success in collecting capillary seepage where it emerges into caves, mainly in sandstone-capped South Dakota caves (Palmer and Palmer, 1989). Slow drips from bedrock pendants were collected in containers held as tightly as possible against the bedrock surface. They did indeed show elevated pH and deficient CO 2 relative to the cave air, but far less than that described in Fig.2. Our procedure was not sophisticated enough to prevent at least some CO 2 exchange with the cave air before pH measurement.
Weathering of cave walls
Where water seeps into the cave in volumes too small to cause visible solution rills, but where conditions are not sufficiently dry for gypsum to precipitate, the bedrock eventually acquires a white weathering rind up to 1.5 cm thick. Thin-section analysis, X-ray diffraction, and chemical analysis of dissolved samples show that the rind simply represents a diminution of crystal size, with no significant mineralogical change except for minor precipitation of silica (described in the next section). Weathering is apparently accompanied by oxidation of organic material, because the gray fog of organics that darkens the unaltered bedrock is absent in the rind and does not appear in the insoluble residue when the rock is digested in dilute HCl. The mass of the organic fraction is too small to measure by gravimetric methods. Except for large residual grains of calcite, most of the rind does not stain with alizarine red, a popular indicator of calcite, giving the false impression that the crust has been altered to a different mineral. The unstained areas are limited almost entirely to the white rind of microcrystalline calcite.
The thickness of the weathering rind varies a great deal, depending on the duration of exposure, moisture conditions, and lithology. The thickest and most distinctive rinds are in the uppermost levels, particularly in porous, granular beds from which there is widespread seepage of capillary moisture. In general the rind thickness decreases downward from the uppermost level ( Collins Avenue ), where the mean thickness is 0.9 cm (maximum of 1.5 cm), to less than 0.2 cm in passages in the St. Louis Limestone (e.g. Martel Avenue ). However, the downward decrease is not uniform, because the thickness depends also on the local rate of capillary seepage. The thickest rinds concentrate around streams and shafts, outside the zone of discrete streams or drips (which, except in highly impure limestones, produce cleanly dissolved surfaces with no weathering rind). In dry upper-level passages, one can predict the proximity of a shaft or vadose stream from the onset or thickening of the weathering rind. On the other hand, the rinds are absent or greatly subdued in areas subject to frequent flooding.
Dissolution within the rinds is apparently fostered by the uptake of CO 2 at the cave walls by water that has previously approached equilibrium with carbonate rocks under closed conditions, as described in the previous section. The amount of water in these areas is not sufficient to produce visible solution features. Instead, dissolution is limited to grain boundaries and cleavage planes within crystals, etching the grains and recrystallizing them to a finer size. Moisture greatly enhances the oxidation of organics. Although much of the dissolved oxygen in infiltrating water is consumed by reaction with organic compounds in the soil, oxygen in the moisture at and near the cave walls is readily replenished by diffusion from the cave air. Bedrock beneath gypsum crusts does not have the white weathering rind, because the amount of moisture necessary to produce rinds is too great to allow gypsum precipitation. Gypsum crusts in Mammoth Cave are derived mainly from the oxidation of pyrite in the bedrock. Sulfuric acid released by this process can form white alteration haloes around the pyrite grains. The absence of rinds beneath gypsum indicates that the wall weathering is not caused by sulfuric acid reactions.
In moist areas, dolomite usually weathers to a white or gray paste. The ceiling of Turner Avenue at the top of Brucker Junction exhibits this pasty texture very well, where the dolomitic base of the Paoli Member of the Girkin Formation is exposed to capillary moisture fed by a nearby drip. The paste consists mainly of an aggregate of solutionally etched dolomite rhombs.
The widespread nature of the rinds suggests that much capillary moisture is fed by diffuse infiltration from the surface. Most such infiltration is concentrated in the neighborhood of discrete stream inputs. Even a vadose stream fed by remote inputs can serve as a source of capillary moisture deep beneath the sandstone cap-rock. For example, capillary seepage wicking into the surrounding limestone from the stream in Waterfall Trail (Crystal Cave), which is perched on the Aux Vases Member of the Ste. Genevieve Limestone more than 45 m above the Green River, has produced a white weathering rind in nearby passages, as well as in the walls and ceiling of the stream passage itself. Kline Trail ("the Crawlway") crosses perpendicularly over Waterfall Trail at an elevation 12 m higher, and the walls of Kline Trail are noticeably weathered in a 50-m zone around the crossing point.
Precipitation of silica by capillary water
Dissolving samples of the white weathering rind in dilute HC1 leaves an insoluble residue that usually includes a small but conspicuous quantity of white non-detrital silica. This was identified as microcrystalline quartz with the aid of X-ray diffraction. It consists of small irregular bodies generally less than a millimeter in diameter, mainly representing pore-lining cement and replacement of certain grains in the bedrock. In oolitic limestones the quartz includes many hollow spherical shells that have replaced certain favorable zones in the oolites. The diameter of individual crystals in the secondary quartz averages about 1-3 mm. This secondary silica occupies only about 2 to 5% of the weathered zone on the average, with a maximum of about 10%, increasing outward toward the cave walls. Typically about half of the total quartz in the weathering rinds consists of detrital grains in the original bedrock.
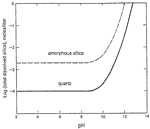
Although the secondary quartz is volumetrically insignificant, its presence helps to confirm the concept of closed-system carbonate dissolution by capillary seepage. Dissolution of silica takes place by hydration to H 4 SiO 4 . Its solubility varies with the type of silica (quartz, opal, etc.). For any given silica type the solubility is nearly constant at pH values below 9 but increases considerably at higher pH owing to dissociation of H 4 SiO 4 to H + and H 3 SiO 4 - (with additional dissociations at even higher pH). This solubility curves are shown in Fig.3. In ordinary karst water the pH rarely exceeds 8.5; but as shown earlier, the pH may exceed 9.0 in small fissures and pores in the vadose zone. Therefore, in these high-pH zones a great deal of silica can be dissolved from chert and quartz and from the weathering of complex silicate minerals, which are available not only in the sandstone but also as impurities in the carbonate rocks. As the water approaches the cave, the pH drops rapidly, and the saturation levels of the various forms of silica rise sharply. Micro crystal line quartz precipitates in the weathering zone at the cave walls. Its precipitation is extremely slow at low temperatures, and measurable quantities are restricted to upper-level passages that have been exposed to weathering for hundreds of thousands of years. The pattern of quartz observed in thin sections tends to mimic that of organic zones in the unaltered bedrock, which may indicate that the quartz has selectively replaced organics.
Fig. 3. Solubility of SiO 2 vs. pH, calculated from thermodynamic values given by Weast and others (1986). Total concentration = H 4 SiO 4 + H 3 SiO 4 - + H 2 SiO 4 = . Solubility of amorphous silica (opal, etc.) is approximate.
Some of insoluble residue in the rinds is indigenous to the bedrock and is not a cave-related precipitate. Comparison of the insoluble percent in the unweathered bedrock with that in the weathering rind for 16 samples having well-developed white weathering rinds shows a mean increase of 2.15 times within the weathered zone. On the average, the quartz content of the sampled weathered zones was 5.75%, compared to 2.92% in the unweathered bedrock. A one-tailed t-test shows more than 99% probability that the weathered zone has a significantly greater insoluble content.
The increase in silica was not caused simply by enrichment of the original detrital fraction in the bedrock by weathering. Linear regression for the data pairs (X,Y), where X = insoluble percent in the unweathered rock and Y = insoluble percent in the weathered zone minus that in the unweathered rock, gives a correlation coefficient of only -0.235, i.e. not only insignificantly low, but negative as well.
A thin detrital dusting of silt-sized quartz (mean diameter 100 µm) is present on the surface of most walls in Mammoth Cave . Some may be air-borne material stirred up by visitors, and some may be residue from flooding. However, most of the silt pre-dates the gypsum crust, since it is disrupted by gypsum wedging, which indicates that it is a natural feature. These observations do not include areas of backflooding, where clay and silt are abundant on the walls. The outer part of the weathering rind containing the detrital coating was removed before the samples were dissolved to obtain the insoluble percentage.
Chert can replace certain zones in the limestone, especially gypsum bodies. Reduction of sulfate in anoxic environments produces an aura of low to moderate pH from the production of hydrogen sulfide and carbon dioxide. Therefore, gypsum and anhydrite are typical targets for silica replacement, especially by high-pH water that has dissolved carbonate rocks in closed systems. Organic-rich material is also commonly replaced for similar reasons. Examples of silica replacement in Mammoth Cave include the nodular dikes in the lower Levias Member of the Ste. Genevieve Limestone (best seen in the Waterfall Trail level of Crystal Cave), and the cauliform quartz nodules in the upper Jl unit of the Joppa Member exposed in the ceiling of Floyd's Lost Passage and parts of New Discovery. These were former anhydrite bodies, as shown by pseudomorphs recognizable under microscopic inspection. Conversion to silica may have little relation to the origin of the cave. In areas far removed from weathering processes, such as bedrock exposed by extensive breakdown in certain caves (e.g. Camp's Gulf Cave in Tennessee), the complete replacement sequence can be seen, from pure anhydrite and gypsum, through partial replacement, to pure silica.
Gypsum replacement of limestone by capillary water
In the driest passages of Mammoth Cave , capillary seepage evaporates at the cave wall and does not form discernable trickles. As CO 2 -depleted water approaches the cave and absorbs CO 2 from the cave air, the CO 3 = content of the water diminishes about 5 times, raising the SO 4 = /CO 3 = ratio accordingly (Palmer, 1986). Besides precipitating gypsum and other evaporite minerals, this water also tends to replace calcite with gypsum in the cave walls.
Replacement of calcite by gypsum can take place by selective grain-by-grain dissolution of calcite, accompanied by simultaneous or subsequent precipitation of gypsum. However, microscopic examination shows that many grains are only partly replaced, and that all their original textures are present. It is uncertain whether this represents mole-for-mole replacement of carbonate by sulfate. Although this mode of replacement ideally causes an increase in volume, the partially replaced grains show no visible expansion. This indicates an open system in which there is a volumetric balance but not a molar balance. Conditions where capillary water emerges into the cave are favorable for this process, regardless of whether the calcite is chemically converted to gypsum or simply replaced. Evaporation increases the concentration of dissolved solids, without which gypsum would be unable to precipitate in this environment. The uptake of CO 2 causes dissolution of the pore walls in limestone, which enhances the Ca ++ content, and the abrupt rise in SO 4 = /CO 3 = promotes the direct replacement of calcite by gypsum.
The replacement of calcite by gypsum involves the following reaction:
![]()
This is an exothermic reaction that is enhanced by lower temperatures. Calculations using thermodynamic values from Weast and others (1986) show that at 12 °C (typical of Mammoth Cave ) the required SO 4 = /CO 3 = ratio must be greater than about 4900 for the reaction to take place. This calculation assumes that the activity of water is 1.0. In air with a relative humidity below 100% the required ratio would be higher, but such conditions are unlikely within the bedrock pores.
Capillary seepage is able to acquire dissolved calcium and sulfate either by oxidation of pyrite or by dissolution of residual gypsum within the bedrock. Both sources are available at Mammoth Cave . However, gypsum crusts and speleothems are concentrated within certain pyrite-rich beds, so it appears that most of the gypsum is derived from pyrite. In many places, such as in Jeanne's Avenue, gypsum flowers emanate from iron oxide blobs that were originally pyrite.
The ability of capillary water to precipitate gypsum at the cave wall shows that it is rich in dissolved sulfate. The solubility of gypsum is about 0.014 moles/liter, but because Ca ++ is limited by the much lower solubility of calcite, sulfate can rise to much higher concentrations, depending on its availability and the rate of calcite precipitation. In the high pH conditions of the closed-system vadose environment, CO 3 = is much more abundant than is usual for karst waters. When the water absorbs CO 2 at the cave wall and the pH drops, the CO 3 = concentration is sharply diminished, causing about a 5-fold boost in SO 4 = /CO 3 = at the cave walls. Regardless of the initial sulfate content, the replacement of calcite is promoted by this change.
White (1976) indicated a similar ratio for the replacement of calcite by gypsum but assumed that the seepage would be degassing at the cave wall, as is true for most vadose water. He predicted that the degassing would cause a decrease in CO 3 = . This sounds feasible, since CO 2 is a potential source of CO 3 = , but in this case it is just the opposite. An increasing CO 2 content causes a drop in pH, accompanied by the conversion of much of the existing CO 3 = to HCO 3 - . The CO 3 = content of the water does in fact decrease where it emerges at the cave wall, but because of an increase of CO 2 in the capillary seepage, rather than degassing.
Most of the major gypsum displays in the cave are located on or just below major pyrite-rich beds, many of them dolomitic limestones. For example, Cleaveland Avenue, New Discovery, and Floyd's Lost Passage are all developed in the same rock unit (Jl unit of the Joppa Member, Ste. Genevieve Limestone), which includes pyrite-rich dolomitic beds. Pyrite is also abundant in the Sample, Aux Vases, Spar Mountain , and Joppa members. Do such beds contain sufficient pyrite to account for the large localized gypsum deposits in the cave? Thin-section measurements of the cross-sectional area of pyrite in unweathered limestone show that pyrite comprises an average of about 0.1% of the original limestone bedrock in the Mammoth Cave area. This figure does not include the large concentrations of pyrite that are common within shaly beds, or relict primary gypsum within the bedrock. A common convention (as with porosity measurements) is to consider the areal percentage equivalent to the volume percentage. The densities of pyrite and gypsum are 5.0 and 2.32 g/cm 3 respectively, and their molecular weights are 119.98 and 172.14 g/mole respectively. One mole of pyrite can produce two moles of gypsum. Therefore, each cubic meter of limestone can ideally produce about 0.006 m 3 of gypsum, and a one-cm-thick gypsum crust could be formed in a passage 2 m in diameter by the oxidation of pyrite in a zone only one meter thick around the passage! The calculations are as follows:
The volume of gypsum per meter of passage = 2prtL = 2p(100)(I)(100) cm 3 = 6.3 X 10 4 cm 3 , where r = passage radius, L = segment length, and t = crust thickness. The volume of bedrock required = V g /(0.001)/(6) = 1.1 X 10 7 cm 3 , where V g volume of gypsum. Expressed as a cylindrical zone around the passage, this volume is equivalent to pL(r b 2 - r p 2 ), where r b = outer radius of weathered bedrock and r p = passage radius. Therefore, the bedrock thickness (r b - r p ) around the passage required to form the gypsum crust is only a little more than 100 cm.
Precipitation of gypsum at the cave walls raises the Mg/Ca ratio, allowing hydromagnesite to precipitate in a few places. Hydromagnesite is present in Thomas Avenue ("Gypsum Route") in Crystal Cave, where seepage is enhanced along the flanks of a sandstone-capped ridge.
Chemical zonation in air-filled passages
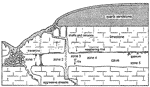
Frequent visitors to caves in the Chester Upland are quite familiar with the lateral changes in moisture conditions and speleothem types that take place in passages that extend from areas of uncapped limestone into areas capped by low-permeability detrital rocks (Fig.4). This zonation is more intricate than generally realized and is greatly influenced by the chemical processes described in this paper. Good examples of chemical zonation can be observed in the main passage of Great Onyx Cave (Edwards Avenue) and in the Kentucky Avenue area, especially near the Frozen Niagara Entrance and New Entrance.
Fig. 4. Generalized profile of a cave passage showing 5 different geochemical zones related to vadose infiltration.
These zones are listed below in the order in which they are encountered as one travels from passages overlain by uncapped limestone to those overlain by a low-permeability cap of detrital rocks:
Zone 1: Water infiltrates through soil-mantled limestone and reaches equilibrium with dissolved limestone at the high CO 2 values acquired in the soil. Degassing of CO 2 takes place in the cave, and if the water is close to saturation the degassing forces calcite to precipitate as speleothems. This is typical of small flows and drips. Larger flows fed by sinkholes generally retain their aggressiveness throughout the cave.
Zone 2: Water infiltrates through the sandstone cap-rock and concentrates as active drips and flows that retain their aggressiveness throughout their entire paths through the cave. The soil in such areas has a Pco 2 about half as great as in zone 1, but the water reaches the cave with little or no dissolved carbonate and is highly aggressive. Some of this water probably achieves calcite saturation under essentially closed-system conditions, just as capillary water does. Uptake of CO 2 from the cave air rejuvenates the aggressiveness.
Zone 3: Capillary water fed by infiltration through the sandstone cap-rock reaches closed-system saturation with calcite, which nearly depletes the CO 2 content. Where the water enters the cave it absorbs CO 2 from the cave atmosphere and becomes aggressive once more, dissolving rills down the cave walls.
Zone 4: Water similar to that of zone 3 enters the cave as diffuse seepage capable of forming only a thin film of moisture on the walls. The water is aggressive but is only able to etch and recrystallize the calcite in the walls to smaller crystal size, forming a white weathering rind. Silica precipitates in some areas owing to the sudden drop in pH. Oxidation of organics helps to account for the light color of the rind. Conditions are too moist for gypsum and other evaporite minerals to form.
Zone 5: Water similar to zone 4 enters the cave at such a small rate that there is no observable moisture film. Evaporation consumes nearly all the moisture. Gypsum and other evaporite minerals are precipitated and gypsum can replace calcite in the crust. The latter process is enhanced by the rapid decrease of CO 3 = at the cave wall, which increases the sulfate/carbonate ratio. Gypsum growth along fractures disrupts the bedrock surface by wedging rock fragments apart and cementing the dislodged fragments in place.
Boundaries between zones migrate with time as the edge of the cap-rock retreats. The dripstone-decorated shaft at Crystal Lake, near the Frozen Niagara Entrance, illustrates the change from aggressive shaft-forming water (zone 2) to supersaturated water depositing speleothems as the result of degassing (zone 1). Shafts farther into the passage still contain solutionally aggressive water. Undersaturated seeps form local rills in the walls of Jeanne's Avenue and Boone Avenue (zone 3). Coarse, granular limestones in many parts of Kentucky Avenue contain fine examples of white weathering rinds (zone 4). Gypsum crusts in the driest parts of these passages represent zone 5, but in places the crust is peeling from the wall as zone 5 changes to zone 4.
Capillary seepage also accounts for other features, such as cave popcorn and aragonite, but these have not been described here, as this type of seepage is generally saturated with high levels of dissolved carbonates before it enters the cave and has a chemistry similar to that of the drips in zone 1. Condensation of moisture on the cave walls has not been investigated in Mammoth Cave, but it is an important agent in weathering and precipitation in many caves in arid and semi-arid climates and may modify the chemistry of capillary seepage even in such a humid environment as Mammoth Cave,
Conclusions
Despite its small volume, capillary seepage has a profound effect on the appearance of Mammoth Cave. It is one of the main controls over the weathering of the limestone walls (including local dissolution, oxidation, and recrystallization), deposition of silica, gypsum, and other evaporite minerals, and replacement of bedrock by gypsum. Recognizing the distinct zonation of capillary seepage in the cave makes it possible to interrelate the chemical processes and moisture conditions within the cave in a systematic way.
References
- Atkinson T.C. 1977. Carbon dioxide in the atmosphere of the unsaturated zone: an important control of ground-water hardness in limestones. Journal of Hydrology 35, 111-123.
- Miotke F.-D. 1975. Der Karst im zentralen Kentucky bei Mammoth Cave: Technical University of Hanover, Germany, Geographical Institute, 360 p.
- Palmer A.N. 1986. Gypsum replacement of limestone by alternating open and closed systems in the vadose zone. Mammoth Cave, Kentucky: Cave Research Foundation Annual Report, p, 27-28.
- Palmer A.N. 1991. Origin and morphology of limestone caves. Geological Society of America Bulletin 103, 1-21.
- Palmer A.N. and Palmer M.V. 1989. Geologic history of the Black Hills caves, South Dakota. National Speleological Society Bulletin, 72-99.
- Weast R.C., Astle W.J. and Beyer W.H. (Eds,). 1986. CRC handbook of Chemistry and Physics. Boca Raton, FL, CRC Press.
- White W.B. 1976. Cave minerals and speleothems. In: Ford T.D. and Cullingford C.H.D. (Eds.): The science of speleology. London: Academic Press, 267-327.
Biographic Sketch
Arthur N. Palmer is professor of hydrology at the State University of New York at Oneonta and director of their Water Resources program. Margaret V. Palmer is a consulting geologist specializing in carbonate petrology. They have spent 30 years studying the geology of the Mammoth Cave System and other caves throughout the world and have written interpretive guidebooks for several National Parks and Monuments, including A Geological Guide to Mammoth Cave National Park, as well as a variety of technical papers on the origin and geology of caves and karst.